If you find these posts interesting and think others might do so, spread the word
Apologies for the lack of posts, but May-June is exam season in the UK University systems, and I’ve been focusing on a couple of other writing projects for the past month, along with a break, in which I mostly avoided the internet.
AZ vindicated?
There’s been some coverage of the AZ vaccine and the risk of blood clots again. A very short report in the Lancet1 seems to underline the very low risk of the thrombosis with thrombocytopenia syndrome (TTS) with the second dose, which backs up the spontaneous reporting data from the MHRA.
The Telegraph (in this thread and an article) make mention of another study suggesting that the risks are no higher with the AstraZeneca Vaccine than the Pfizer vaccine.

Just last week, new research scotched damning claims AstraZeneca posed a blood clot risk. A team of researchers from Spain, the UK and the Netherlands compared data from more than 1.3million people and concluded that those who had the UK jab developed blood clots at the same rate as those who had the far more expensive Pfizer/BioNTech.
More importantly, they found people who had Covid-19 developed blood clots at a far higher rate than those who received neither vaccine.
This appears to be from a Lancet preprint, and appears to be accurate reporting.2 The study compared rates of thromboembolism and thrombocytopaenia with the Pfizer vaccine and the AstraZeneca vaccine, and used a COVID-19 diagnosis as a comparator - highlighting risks of vaccines without the contextual risks for the disease are inherently misleading. They found similar rates for clots with both vaccines, but COVID-19 was far more risky for clots than either. Obviously, this contradicts the spontaneously reported data in the UK and Europe, where the signal of thrombosis with thrombocytopenia syndrome (TTS) is only of concern with the AstraZeneca vaccine. Why is this?
This is an observational study using a primary care database in one area of Spain. As the authors point out:
thromboembolic events were identified using primary care records, and therefore they might be underestimated in all cohorts. For instance, we were unable to identify particularly rare events of special interest, such as CVST or SVT. Although underestimation of events was likely non-differential across cohorts for events before March 2021, we cannot exclude that detection bias following the EMA’s signal alert report might have led to differential measurement error between the vaccinated and comparator cohorts from March onwards.
If the primary care records did not record cases well, then that might explain why the risk thrombotic events might not be different between the vaccines. So, while this is an interesting study using a decent database (from good researchers), and adds some useful data to the mix, I doubt it will have any impact on the decisions already made by the MHRA and EMA with regard to the risks of TTS (and the communications of risk to the public). Although case reports (which is what the spontaneous reports to the Yellow Card scheme are) have their deficiencies, for serious adverse reactions like TTS I’m reasonably confident that the majority of cases are being reported (at least since March when it became widely reported). Any case of TTS occurring in the UK is going to be screened to look for any potential vaccine involvement.
I am not convinced there is a good argument to be made that cases of TTS occurring after the use of the Pfizer vaccine would be preferentially un-reported to the MHRA or EMA, which would explain the difference between the spontaneous reports versus this new study. So, I’m still of the opinion that there is a small risk of TTS with the AZ vaccine that appears far less likely with the Pfizer vaccine. Strong signals in spontaneous data, for all the limitations of such data, need fairly strong counter-evidence to be ignored. I don’t think this study does that.
It does highlight, again, that both the AstraZeneca and Pfizer vaccine are far safer in terms of blood clots than COVID-19. So if you haven’t had the vaccine yet, do get it at the earliest available opportunity.
No prescription, no harms?
There’s an interesting scoping review at Drug Safety on the non-medical and extra-medical uses of Non-steroidal anti-inflammatory drugs (NSAIDs). 3 Sports use comes up a lot as you might expect, including a study I was involved in, but this point stands out…
A common feature across many of the studies was the widely held belief that because a product can be obtained without a prescription and purchased OTC, it does not pose any significant threat to health.
To be fair, the whole class of over-the-counter (OTC) medicines exists for drugs that are considered fairly safe to be used outside of a prescribing setting, so this is perhaps understandable. Perhaps the actual problem is that NSAIDs are inherently risky for OTC drugs.
Change of focus when it comes to risk
I met with a friend at weekend in a full Mexican restaurant, with a live singer, and then went on to a packed bar for a couple of hours. I was trying to work out whether the 20 mile round trip drive was more risky than the 4 hours in an enclosed space. I’ve come to realise on reflection, a few weeks after vaccination, that at a personal level I have defaulted to factoring in COVID-19 as a background risk like any other respiratory infection.
This isn’t to say I’m not concerned about wider societal issues like the immunocompromised, or pressures on the NHS if cases really surged, but the risk of COVID-19 to me personally, based on the effectiveness of the vaccines, is something I am willing to accept without consciously over-thinking every situation.
This comes as somewhat of a surprise to me, since I was pretty much a zealot at the start of the pandemic, including using a mask since the 16th of March 2020 (I remember getting annoyed at a blood donation centre when they would not admit me with a mask). I didn’t stop hand wiping my food shopping down until late summer 2020 and hand gelled constantly. I stopped the latter two behaviours after reading this paper that seemed to indicate surfaces weren't the issue (plus washing your shopping is a nightmare, and my hands were sore).
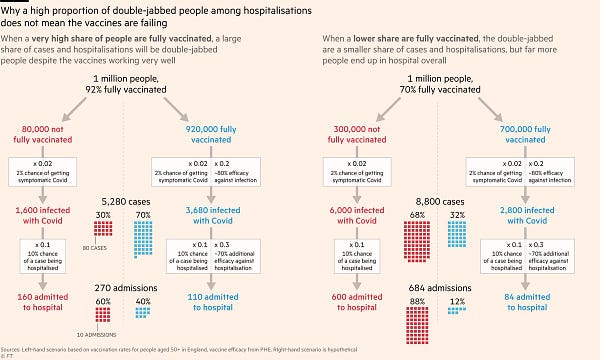
I suppose it will take time for people to re-adjust their risk ‘thermostat’, but I am not convinced that some of the information environments we create for ourselves with social media help with this process. Nor does some of the reporting about breakthrough infections in the vaccinated.

The reason why this is misleading is excellently illustrated here by John Burn-Murdoch:

That’s all and stay safe.
Anthony
Don’t forget to report your suspected adverse effects from medicines and vaccines. In the UK, this means using the Yellow Card Scheme.
Bhuyan, Prakash et al.Very rare thrombosis with thrombocytopenia after second AZD1222 dose: a global safety database analysis. Lancet DOI: https://doi.org/10.1016/S0140-6736(21)01693-7
Burn, Edward and Roel, Elena and Pistillo, Andrea and Fernandez-Bertolín, Sergio and Aragón, Maria and Reyes, Carlen and Verhamme, Katia and Rijnbeek, Peter and Li, Xintong and Strauss, Victoria and Prieto-Alhambra, Daniel and Duarte-Salles, Talita, Thromboembolic Events and Thrombosis With Thrombocytopenia After COVID-19 Infection and Vaccination in Catalonia, Spain. Available at SSRN: https://ssrn.com/abstract=3886421 or http://dx.doi.org/10.2139/ssrn.3886421
Brennan, R., Wazaify, M., Shawabkeh, H. et al. A Scoping Review of Non-Medical and Extra-Medical Use of Non-Steroidal Anti-Inflammatory Drugs (NSAIDs). Drug Saf (2021). https://doi.org/10.1007/s40264-021-01085-9


