Is it Safe? Pushing vaccines in a time of low prevalence
And why the vaccine IP debate is 'thought and prayers'.
If you find these posts interesting and think others might do so, spread the word
It’s now a month since the last major ‘course correction’ on vaccination in the UK, where the JCVI carried made it preferable for the under 30s to have a vaccine other than the AstraZeneca Vaccine. This week Prof Vam Tam, Dr June Raine (MHRA), and Prof Wei Shen Lim (JCVI) carried out a new press conference.
The major outcome of this is that the AstraZeneca vaccine is not the preferred vaccine for those under 40 years of age. This is largely due to the fall in risk of COVID-19 as the prevalence of COVID-19 falls as a result of both vaccine roll-out and the UK lockdown. This means that the benefit-harm becomes more ‘finely balanced” for the AstraZeneca vaccine for younger age groups, where risk seems to be 1 in 60,000 for the thrombotic . For those under 40 who are due a second dose of AstraZeneca vaccine, the risk is extremely low. None of the few cases reported after a second dose of AstraZeneca vaccine appear to be linked to the anti-PF4 anti-bodies, which is thought to be the immune reaction to the vaccine causing the thrombocytopaenia and thrombosis.
For the past few weeks I have been complaining that the use of the precautionary principle in pausing the J&J vaccine and the AstraZeneca vaccine was wrong-headed, particularly in countries with high prevalence of COVID-19. Does the emerging data on the thrombosis linked to thrombocytopaenia, and the JCVI preference for alternative vaccines in the under 30s (April 7th) and under 40s (May 7th) mean I was wrong to hold this opinion? It’s always worth re-visiting things.
Making adjustments on the basis of emergent evidence seems like a sensible position. When I was suggesting pauses in France and Germany were not sensible, they had significant COVID-19 cases which dwarfed any know risks from the AstraZeneca vaccine. They weren’t Norway (were the risk of COVID-19 was much smaller, a total of 767 deaths since the start of the pandemic). Taking extreme examples, in New Zealand with minimal COVID-19 cases and great healthcare, then the risk of a vaccine would obviously be higher than the benefits (assuming New Zealand could maintain indefinitely this situation), and in India with rising cases and deaths, and a health service that has been overwhelmed the benefits of the vaccine clearly are higher than the harms.
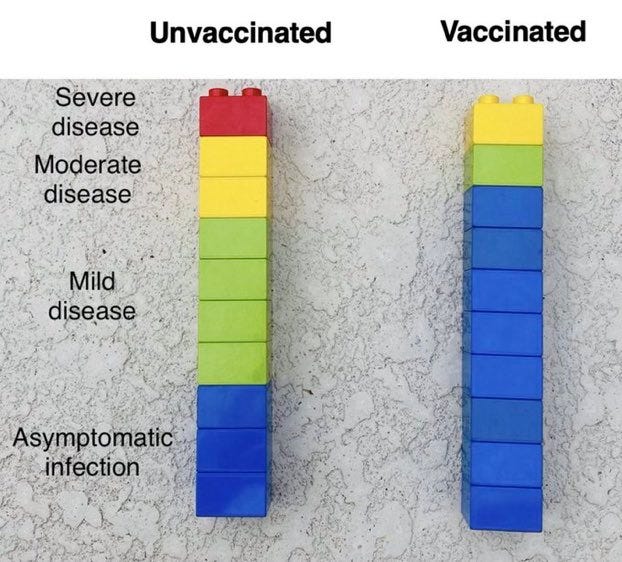
The tricky bit is the transition from high COVID-19 prevalence to low COVID-19 prevalence, and maintaining public confidence in the vaccine as the risk of the disease become attenuated. That’s what is wrong with this graphic. It doesn't include prevalence of disease. If you know you are going to be infected with COVID-19, then it is clear the vaccine is the right calculation.
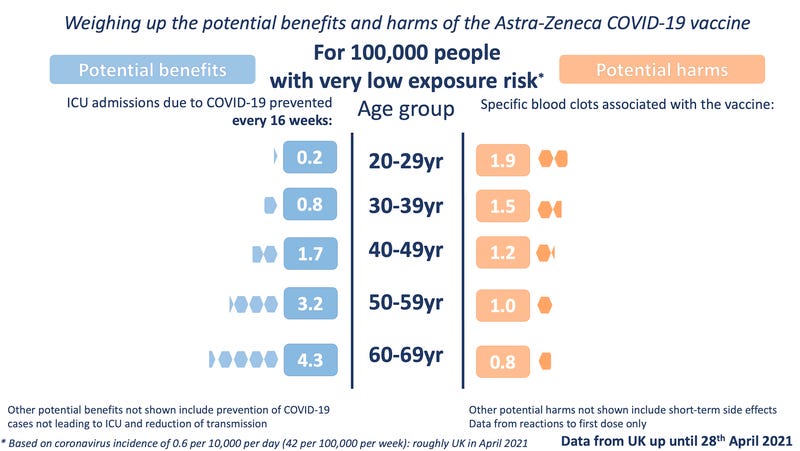
The Winton Centre for Risk and Evidence Communication have a far better set of graphics, including a very low prevalence version which makes the under 40s decision of the JCVI clear.
Having a vaccine roll-out during a peak of the disease hits a sweet spot, and lets you build up momentum. The UK is now at 67.2% first dose, and 33.5% second dose. Only now are we starting to hit the problem of convincing people to have a vaccine in the face of low disease prevalence. Even then, for many people it isn’t just about personal risk, but also the wider benefits of the vaccine roll-out in terms of opening up society, and a return to a form of life that is perhaps more like before. Issues like travel also factor. So, it isn’t just about balancing fairly abstract risks.
For some countries, they are starting their campaigns with low prevalence of COVID-19, this is turning into a problem.
Less than half of people aged over 50 are willing to get the AstraZeneca vaccine despite it being recommended by Australia’s health officials, according to the latest Guardian Essential poll which found confidence in the rollout strategy is waning.
The survey of 1,090 respondents, taken amid a fresh political furore over hotel quarantine and after weeks of confusion about the government’s vaccine rollout, has also tracked a significant increase in those who said they would never get vaccinated against Covid-19, up from 12% to 16% over the past six weeks.
This makes sense if you look at the 3C model of vaccine hesitancy1:
Vaccination complacency exists where perceived risks of vaccine-preventable diseases are low and vaccination is not deemed a necessary preventive action. Complacency about a particular vaccine or about vaccination in general is influenced by many factors, including other life/health responsibilities that may be seen to be more important at that point in time. Immunization programme success may, paradoxically, result in complacency and ultimately, hesitancy, as individuals weigh risks of vaccination with a particular vaccine against risks of the disease the vaccine prevents that disease is no longer common.
One of those paradoxes of life!
It will be interesting to see how the different ways of handling COVID-19 pan out in the long term.
One of the other thing happening recently has been the push, and US support for, the loss of IP protection on vaccines. This has been a constant refrain on social media, and is one of seemingly simple cheap fixes. The Apple computer I’m writing this on has IP protection, but if you told me the IP protection was gone it would still take time for to tool up factories and get production rolling. And would they be made to the same quality? For vaccines this is worse, it is an amazingly complex process with incredibly hard logistics. I have a lot of sympathy with this.

The issue is production capacity, not IP. A major push on investment in production facilities is needed, and that costs money. The IP decision costs nothing, but creates no capacity. This is not like the 1990s IP fight over HIV medication.
Pfizer’s CEO has recently posted about this. They risked over $2 billion developing their vaccine, and have concerns about a possible scramble for raw materials that could endanger production.
Currently, infrastructure is not the bottleneck for us manufacturing faster. The restriction is the scarcity of highly specialized raw materials needed to produce our vaccine. These 280 different materials or components are produced by many suppliers in 19 different countries. Many of them needed our substantial support (technical and financial) to ramp up their production. Right now, virtually every single gram of raw material produced is shipped immediately into our manufacturing facilities and is converted immediately and reliably to vaccines that are shipped immediately around the world (91 countries to date.) The proposed waiver for COVID-19 vaccines, threatens to disrupt the flow of raw materials. It will unleash a scramble for the critical inputs we require in order to make a safe and effective vaccine. Entities with little or no experience in manufacturing vaccines are likely to chase the very raw materials we require to scale our production, putting the safety and security of all at risk.
Of course he would say this, but the case that this is about production capacity, not IP stands. The EU are also skeptical about this.
Other articles on this:
Waiving IP by Derek Lowe.
Patents are Not the Problem! Marginal Revolution
Waiving the vaccine IP is a huge blunder by Tom Chivers
Tom says it well
Saying you’ll waive IP rights is cheap in the worst sense of the word. It gets headlines and approving tweets but it won’t do much good and might do lots of harm. Instead: spend the money. Build factories. Give hundreds of billions of dollars of rich western taxpayers’ money to Covax. Stop trying to push the risk, cost and blame onto the pharma industry.
That’s all and stay safe.
Anthony
Don’t forget to report your suspected adverse effects from medicines and vaccines. In the UK, this means using the Yellow Card Scheme.
MacDonald NE; SAGE Working Group on Vaccine Hesitancy. Vaccine hesitancy: Definition, scope and determinants. Vaccine. 2015 Aug 14;33(34):4161-4. doi: 10.1016/j.vaccine.2015.04.036. Epub 2015 Apr 17. PMID: 25896383.